- Your cart is empty
- Continue Shopping
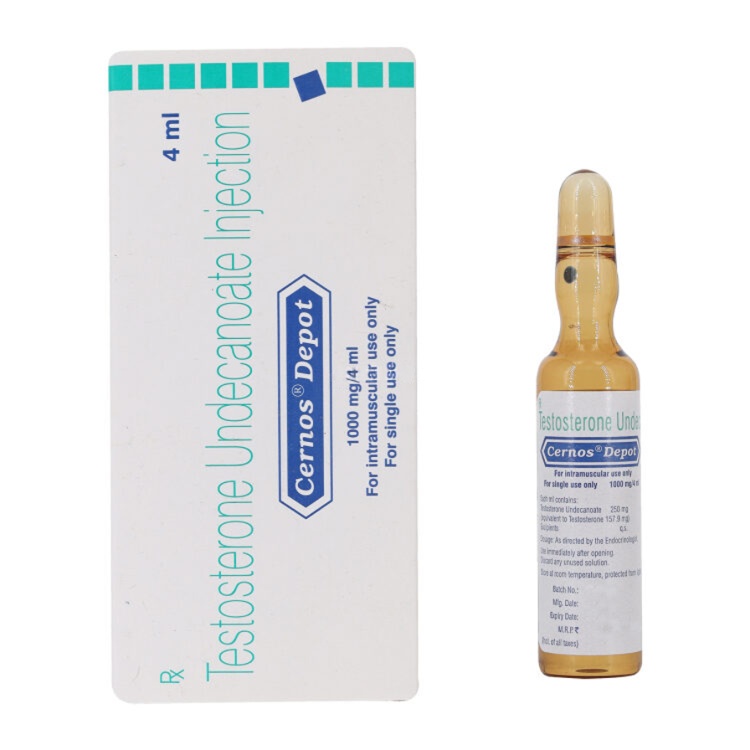
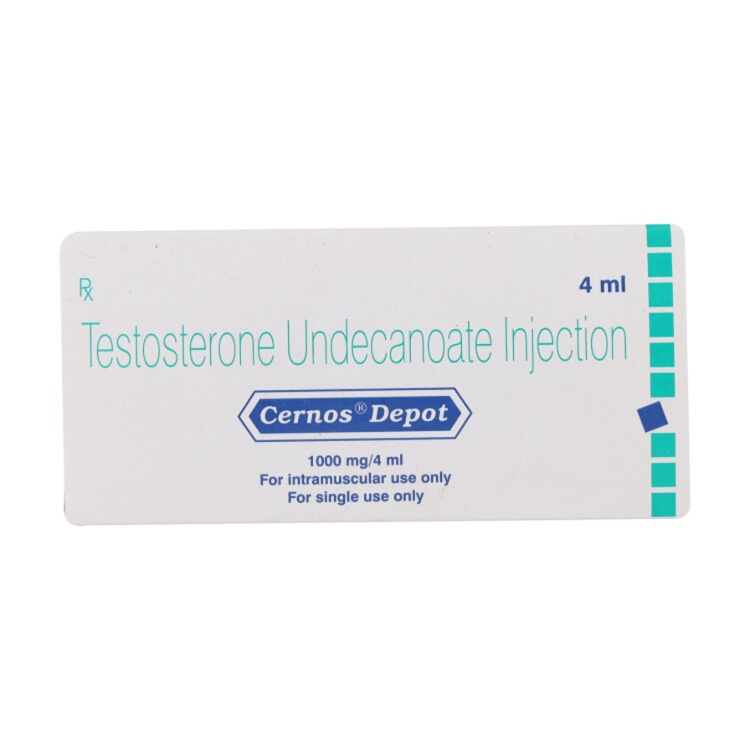
Cernos Depot 1000mg/4ml
$106.20
Cernos Depot (Testosterone Undecanoate) is a long-acting injectable testosterone replacement therapy for treating male hypogonadism and low testosterone levels. This pharmaceutical-grade formulation provides sustained hormone release for 10-14 weeks, effectively increasing muscle mass, enhancing libido, improving energy levels, and supporting overall male health. Professional administration required with regular medical monitoring.
996 in stock
Cernos Depot (Testosterone Undecanoate) – Complete Product Information
Description
Cernos Depot Injection contains Testosterone Undecanoate, a long-acting ester of the primary male sex hormone. This pharmaceutical-grade hormone is vital for the development of male characteristics, enhancing muscle growth, stimulating libido, supporting fertility, and maintaining overall male health.
Testosterone Undecanoate is prescribed to address disorders stemming from low testosterone levels in males, including late-onset puberty, hypogonadism, impotence, osteoporosis, and erectile dysfunction (the inability to achieve or sustain an erection suitable for sexual activity). It functions by replenishing testosterone to normal physiological levels through sustained release technology.
Potential Side Effects
Common side effects of Cernos Depot Injection may include acne, hair thinning, increased body weight, insomnia, headaches, nausea, vomiting, and skin flushing. Typically, these reactions are short-lived and subside independently. Please contact your physician if side effects continue or become more severe.
Administration Guidelines
Administration of Cernos Depot Injection must be performed by a trained healthcare professional in a medical setting; self-administration is strictly prohibited. Your doctor will determine the correct dosage and treatment timeline according to your specific health status and testosterone levels.
Contraindications
Cernos Depot Injection is contraindicated in cases of known hypersensitivity to testosterone undecanoate or its components, or for patients diagnosed with prostate cancer or male breast cancer. Disclose all current medications and health issues to your doctor to ensure this treatment is appropriate for you. Exercise caution when using this medicine if you have cardiac, hepatic, or renal impairments. This injection is not for use in female patients.
Cernos Depot Side Effects
Primary and Secondary Adverse Reactions:
- Gynecomastia (breast tissue enlargement in males)
- Weight gain and increased body mass
- Skin acne and oily skin
- Alopecia (hair loss or thinning)
- Sleep disturbances and insomnia
- Altered heart rhythm (tachycardia, bradycardia, or arrhythmia)
- Injection site discomfort or pain
- Headache and dizziness
- Nausea or vomiting
- Mood changes or increased aggression
Medical Uses of Cernos Depot
Primary Indications:
Male Hypogonadism (Testosterone Deficiency)
Hypogonadism refers to inadequate testosterone production by the testes due to impaired testicular function. Cernos Depot Injection is utilized to manage this condition through testosterone replacement therapy (TRT), which can address symptoms including:
- Reduced bone density and osteoporosis risk
- Diminished muscle mass and strength
- Erectile dysfunction and low libido
- Male infertility and reduced sperm production
- Delayed puberty in adolescent males
- Chronic fatigue and decreased energy levels
- Depression and mood disorders related to low testosterone
Pharmacokinetics
Onset of Action: Testosterone Undecanoate provides gradual release with therapeutic effects typically observed within 2-4 weeks of administration.
Duration of Action: The long-acting ester formulation provides sustained testosterone levels for 10-14 weeks, making it suitable for less frequent dosing compared to shorter-acting testosterone preparations.
Metabolism: Metabolized primarily in the liver to active and inactive metabolites.
Elimination: Excreted through urine and feces.
Cernos Depot Additional Safety Information
Alcohol Interaction
The interaction between alcohol and testosterone undecanoate is not extensively documented. Moderate to heavy alcohol consumption may affect liver function and hormone metabolism. Consultation with your doctor prior to alcohol consumption is recommended, especially during treatment initiation.
Dependency Potential
Cernos Depot may carry a risk of psychological dependency with prolonged use, particularly if used for non-medical purposes or at supraphysiological doses. Always follow prescribed dosing protocols.
Cernos Depot Use in Women
This medicine is not designed for use in women and is contraindicated during pregnancy and lactation due to potential virilization effects on female patients and developing fetuses.
Cernos Depot Contraindications (When Not to Use)
Absolute Contraindications:
Allergy/Hypersensitivity: Do not use if allergic to Testosterone Undecanoate, other testosterone esters, or any excipient. Seek urgent medical care for signs of hypersensitivity such as rash, facial/throat swelling, severe dizziness, or breathing difficulties.
Male Breast Cancer: Contraindicated in patients with cancer of the male breast tissue, as androgens may stimulate tumor growth.
Prostate Cancer: Not recommended for individuals with current or past diagnosis of prostate cancer or suspected prostate malignancy, as testosterone may exacerbate disease progression.
Severe Cardiac Insufficiency: Patients with severe heart failure should avoid this medication.
Severe Hepatic Impairment: Not recommended for patients with serious liver disease.
Special Warnings & Precautions
For Specific Populations:
Pregnancy & Breast-feeding: Not indicated for female use due to androgenic effects and potential harm to developing fetuses.
General Precautions:
Cardiovascular Conditions: Use with caution in patients with heart disease, hypertension, or history of myocardial infarction or stroke. Testosterone therapy can elevate blood pressure, increase hematocrit levels, and potentially raise cardiovascular risk. Regular cardiac monitoring and blood pressure checks may be advised.
Polycythemia (Elevated Red Blood Cell Count): Caution is needed in patients with polycythemia, as testosterone stimulates erythropoiesis (red blood cell production), potentially worsening this condition. Regular monitoring of hemoglobin and hematocrit levels is recommended, with dose adjustment or temporary discontinuation if levels become excessive.
Hypercalcemia: Testosterone may reduce calcium excretion and increase serum calcium levels. Use cautiously in patients with hypercalcemia, renal impairment, or hyperparathyroidism. Regular serum calcium monitoring may be necessary.
Sleep Apnea: Testosterone therapy may worsen obstructive sleep apnea, particularly in obese patients or those with existing respiratory conditions.
Prostate Health Monitoring: Regular prostate-specific antigen (PSA) testing and digital rectal examinations are recommended for men over 40 or those with prostate risk factors.
Substance Abuse & Misuse: Avoid exceeding prescribed doses or extending treatment duration beyond medical recommendations to prevent potential misuse, abuse, or adverse effects associated with supraphysiological testosterone levels.
Intended Gender: For use in male patients only.
Dosage Information
Administration Schedule:
Dosing is individualized based on testosterone levels, clinical response, and patient tolerance. Typical administration ranges from every 10-14 weeks for maintenance therapy.
Missed Dose: Unlikely to occur, as administration is controlled and scheduled by a healthcare provider. Contact your clinic if you miss a scheduled appointment.
Overdose: Risk is minimal under proper medical supervision. Acute overdose may manifest as excessive androgenic effects. Treatment is supportive and symptom-based, managed by qualified clinicians.
Drug & Disease Interactions
Significant Drug Interactions:
Anticoagulants (Warfarin): Testosterone may enhance anticoagulant effects, increasing bleeding risk. More frequent INR monitoring is required.
Antidiabetic Medications (Insulin, Glimepiride): May improve insulin sensitivity and lower blood glucose. Dose adjustments of diabetes medications may be necessary.
Thyroid Hormones (Levothyroxine): Testosterone may alter thyroid hormone binding, potentially requiring levothyroxine dose adjustment.
Corticosteroids: Concurrent use may increase fluid retention risk.
Azole Antifungals (Fluconazole): May affect testosterone metabolism.
Always provide your doctor with a complete list of all medications, supplements, and herbal products you are taking.
Disease Interactions:
Fluid Retention/Edema: Testosterone can promote sodium and water retention, potentially worsening edema and cardiovascular strain in susceptible patients.
Bleeding Disorders: May affect coagulation factors and elevate bleeding risk in patients with hemophilia or other clotting disorders.
Diabetes Mellitus: Can affect glycemic control and insulin sensitivity. Frequent blood glucose monitoring is advised, with possible adjustment of antidiabetic therapy.
Hepatic Impairment: Use cautiously in liver disease, as impaired metabolism can lead to drug accumulation. Regular liver function monitoring is recommended.
Renal Impairment: Caution advised in kidney disease due to altered fluid balance and potential electrolyte disturbances. Renal function monitoring may be required.
Benign Prostatic Hyperplasia (BPH): May worsen urinary symptoms in men with enlarged prostate.
Cernos Depot Administration Instructions
Cernos Depot Injection must be administered only by a qualified healthcare professional in a clinical environment via deep intramuscular injection. The injection should be given slowly due to the oil-based formulation.
Patient Responsibilities:
- Report any persistent or worsening symptoms to your doctor immediately
- Maintain regular follow-up appointments and laboratory monitoring throughout therapy
- Provide full disclosure of medical history and concurrent medications
- Attend scheduled PSA and hematocrit monitoring
Additional Clinical Details
Timing with Meals: Administration is not dependent on meals or fasting status.
Effect on Alertness: Does not typically induce drowsiness or impair cognitive function. However, individual responses may vary.
Mechanism of Action: Testosterone Undecanoate is a prodrug that is converted to testosterone in the body. It exerts therapeutic effects by binding to androgen receptors, correcting testosterone deficiency and alleviating symptoms of hormonal imbalance. Effects include:
- Increased protein synthesis and muscle mass
- Enhanced bone mineral density
- Improved libido and sexual function
- Stimulation of erythropoiesis
- Maintenance of secondary male characteristics
Drug Classification: Androgen and anabolic steroid (Schedule III controlled substance in some jurisdictions)
Storage: Store at room temperature, protected from light and moisture. Keep out of reach of children.
Monitoring Requirements During Therapy
Regular monitoring should include:
- Serum testosterone levels
- Complete blood count (hemoglobin, hematocrit)
- Prostate-specific antigen (PSA)
- Liver function tests
- Lipid profile
- Blood pressure
- Bone density (in osteoporosis cases)
- Blood glucose (in diabetic patients)
| Brands | |
|---|---|
| Use | |
| Form | |
| Dose | |
| Size | |
| Shipping Zones |